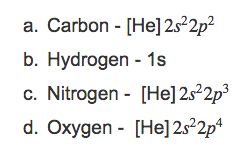Formula for Acetaminophen:
Acetaminophen is made up of four elements that can be found on the periodic table of elements. Those four elements are, Carbon, Hydrogen, Nitrogen, and Oxygen.
Composition
Carbon: 63.56%
Hydrogen: 6%
Nitrogen: 9.27%
Oxygen: 21.17%
Mass: 151.16 g/mol
Hydrogen: 6%
Nitrogen: 9.27%
Oxygen: 21.17%
Mass: 151.16 g/mol
Noble Gas Notation of Elements:
Lewis Diagram of Acetaminophen: |
Skeletal Diagram: |
Lewis Diagrams show the bond between two electrons. The dots around the atoms on a Lewis diagram show the number of valence electrons in an atom. The outer shell wants to be filled with valence electrons in order to be as stable as possible. My diagram shows which atoms are being bonded and which how many electrons are being shared. Acetaminophen is covalently bonded as can be seen in the diagram above. In a covalent bond electrons are shared between two atoms. The diagram on the right is a skeletal diagram which shows the bond without the valence electrons. There are two types of bonds that are apparent in acetaminophen: single and double. A single bond is when one valence electron is being shared on each side. A double bond is when two electrons from each atom are being shared.